Bayer's Nubeqa secures expanded access to UK market for prostate cancer treatment | HealthCare Middle East & Africa Magazine

Logo du Groupe Bayer à Paris, France, sur 16 mai 2020. Groupe pharmaceutique Bayer annonce un nouveau médicament - Nubeqa - contre le cancer de la prostate. (Photo de Daniel Pier/NurPhoto Photo Stock - Alamy

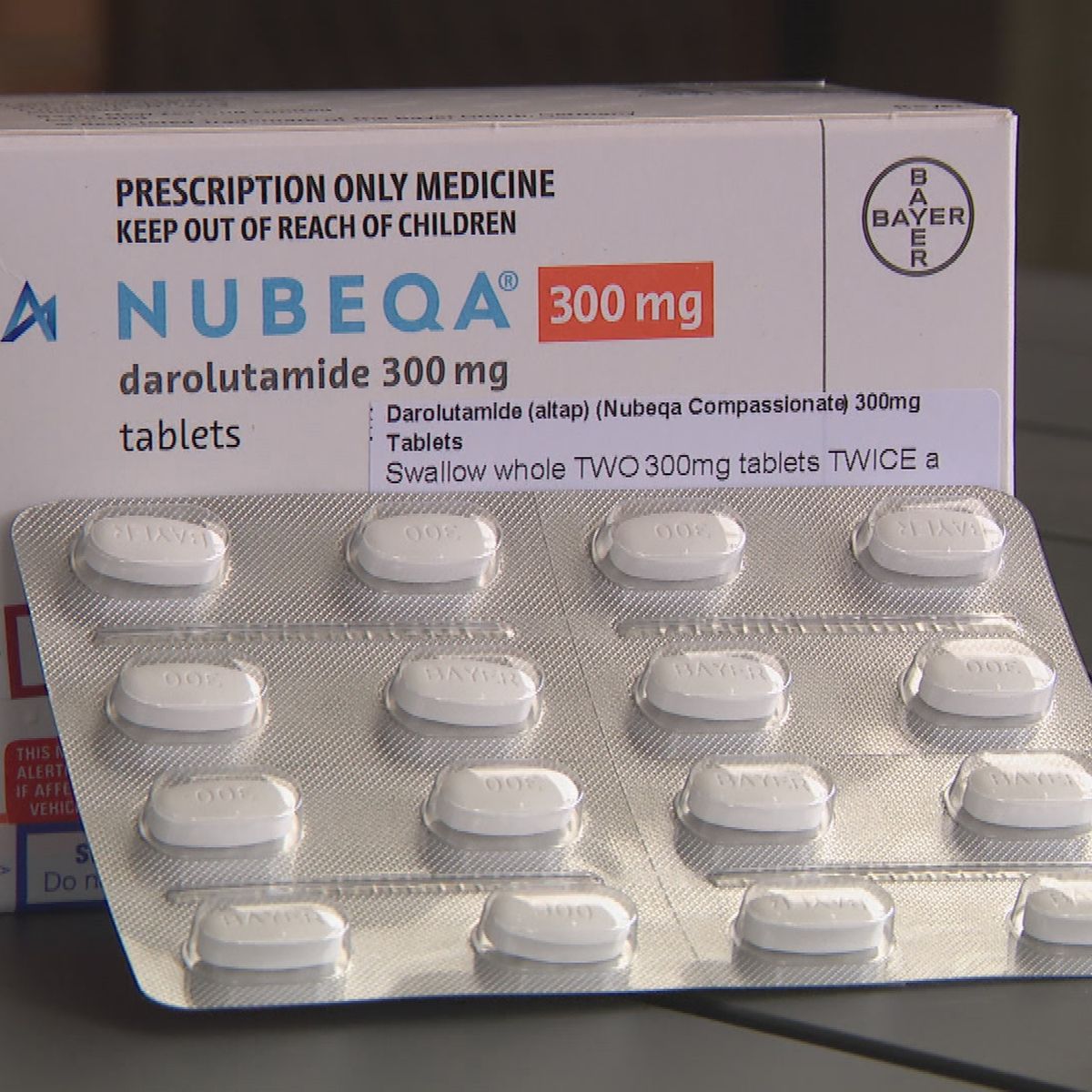
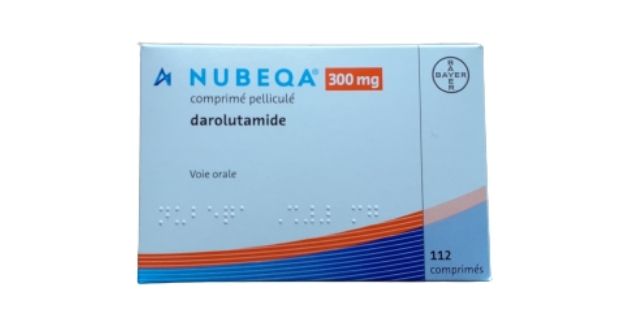
FDA approves Bayer's Nubeqa® (darolutamide), a new treatment for men with non-metastatic castration-resistant prostate cancer