Construction of β-Amino Sulfones from Sodium Metabisulfite via a Radical 1,4-Amino Migration | Organic Letters

A solution of 12 in aqueous KI on being treated with an aqueous solution of Na2S2O3 gets decolourised. The reaction in question is: (a) Na2S2O3 + H2O +12 —> Lodometni stration Na2S204 +

Synthetic schemes for HyP-2 and HyP-2C (control compound; no o-MOM).... | Download Scientific Diagram

Construction of β-Amino Sulfones from Sodium Metabisulfite via a Radical 1,4-Amino Migration | Organic Letters

SOLVED: Name Section When sodium metabisulfite (Na2S2O5) reacts with potassium iodate (KIO3), one of the products of the reaction is elemental iodine (I2). What is the normal oxidation state (ionic charge) of

SOLVED: Sulfur dioxide can be produced in the laboratory by the reaction of hydrochloric acid, HCl, and a sulfite salt such as sodium sulfite. The unbalanced reaction is shown below: HCl (g) +

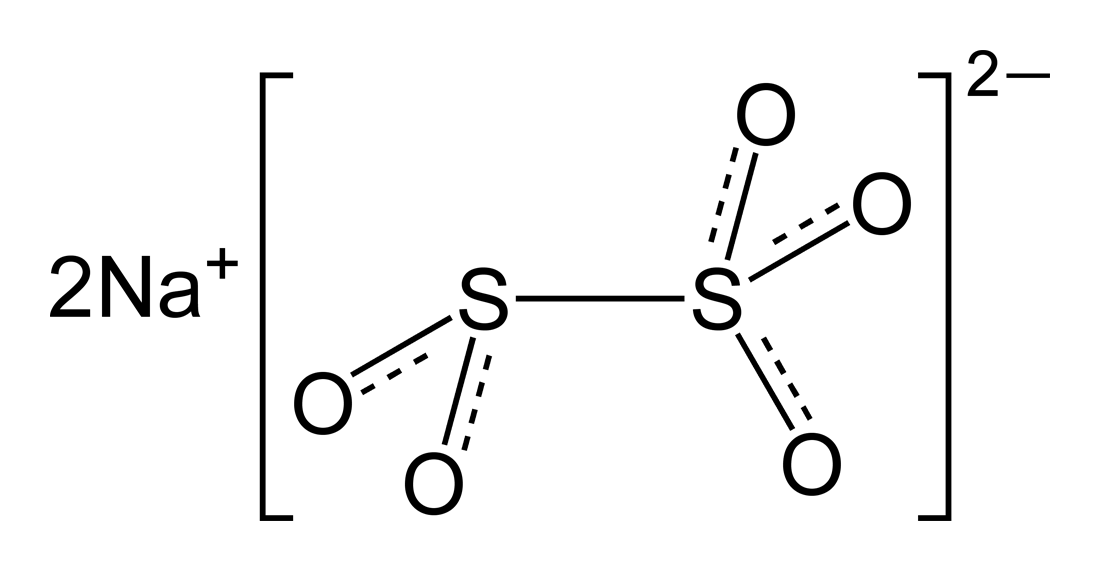
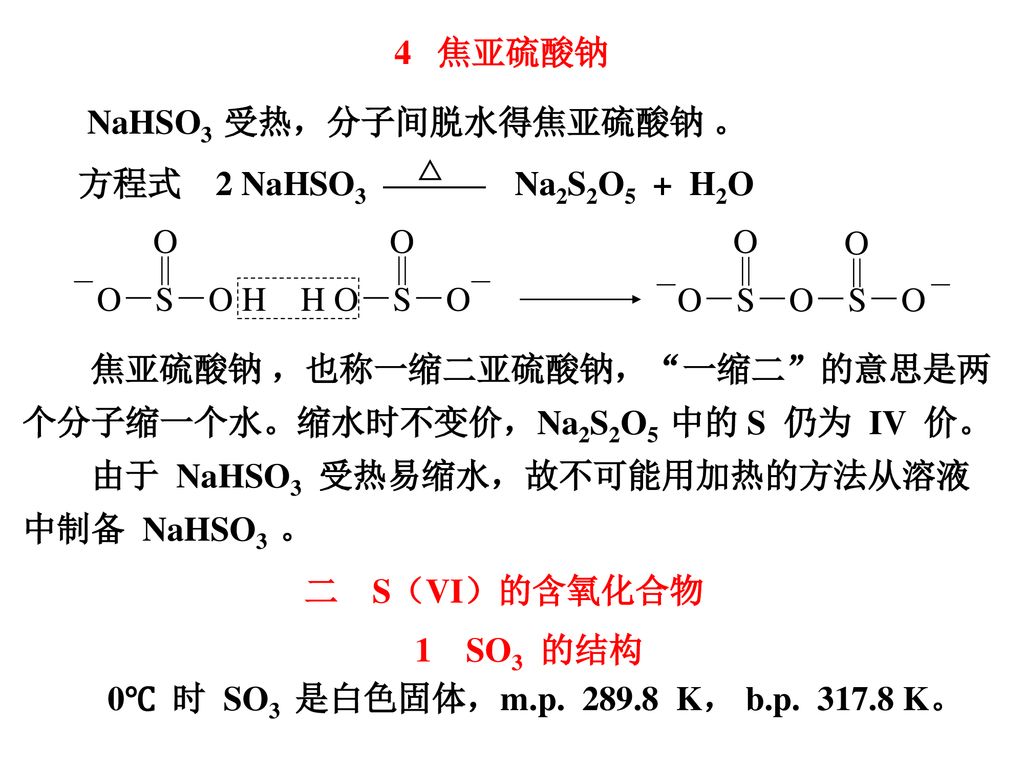
SOLVED: In the lab, you generated bisulfite ion (HSO3-) from a solution of sodium metabisulfite (Na2S2O5) according to the following net ionic equation: S2O5 (aq) + H2O (l) â†' 2 HSO3- (aq)
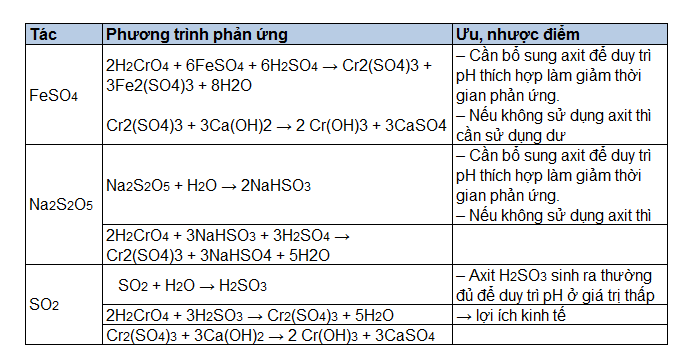
A Comparison of the Use of Sodium Metabisulfite and Sodium Dithionite for Removing Rust Stains from Paper

The synthetic protocol of the compounds. Reagents and conditions; i:... | Download Scientific Diagram