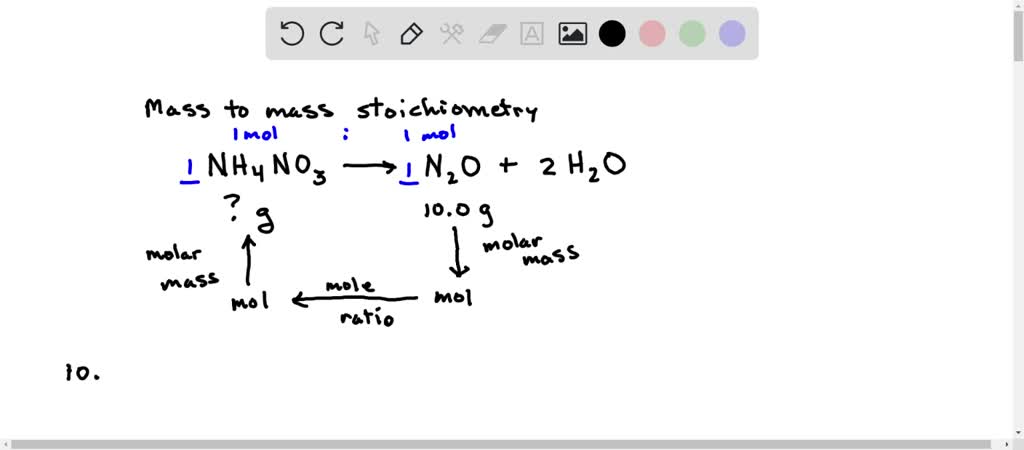
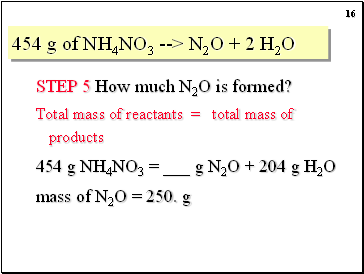
SOLVED: Considering the following balanced equation. NH4NO3 (s) → N2O (g) + 2 H2O (g) Calculate the mass (g) of NH4NO3 needed to produce 10.0 g of nitrous oxide. The molar masses

SOLVED: 1. Al + O2 â†' Al2O3 2. Na + H2O â†' NaOH + H2 3. NH3 + O2 â†' N2O + H2O 4. PbO + NH3 â†' Pb + N2 +
The molar heat of formation of NH4NO3 (s) is -367.57 kJ and those of N2O(g), H2O(l) are 81.46 - Sarthaks eConnect | Largest Online Education Community

The Isotopocule values (SPN2O and δ¹⁸ON2O/H2O) of soil‐borne N2O from... | Download Scientific Diagram

Figure 1 from Structure and Abundance of Nitrous Oxide Complexes in Earth's Atmosphere. | Semantic Scholar








![Gujrati] Balance the equations Mg + NO3^(-) to Mg^(2+) + N2O + H2O) Gujrati] Balance the equations Mg + NO3^(-) to Mg^(2+) + N2O + H2O)](https://static.doubtnut.com/ss/web-overlay-thumb/5987293.webp)





