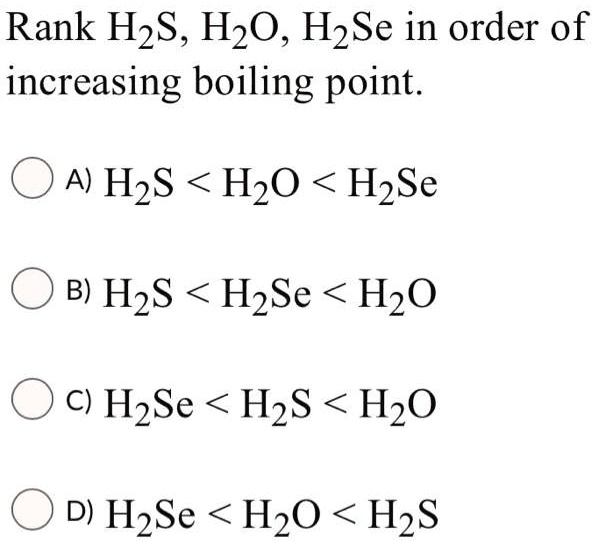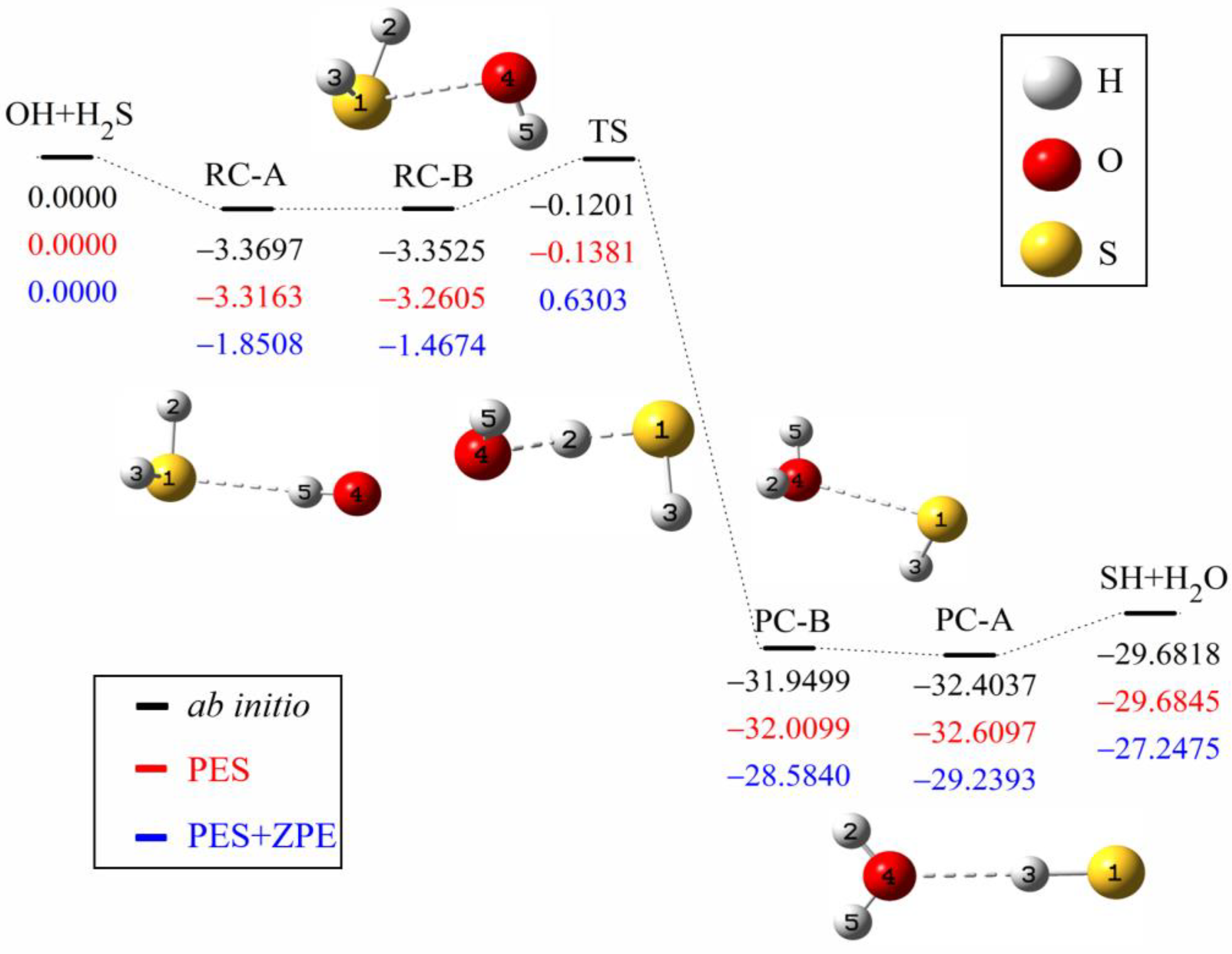
Symmetry | Free Full-Text | Quasi-Classical Trajectory Dynamics Study of the Reaction OH + H2S→H2O + SH and Its Isotopic Variants: Comparison with Experiment

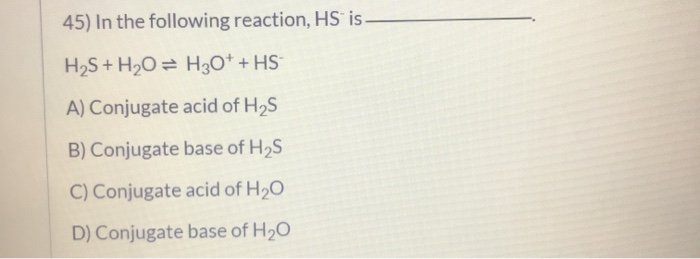
SOLVED: QUESTION 11 Is H2O or H2S more nucleophilic in protic solvent, and why? H2S is more nucleophilic because it is less solvated by the protic solvent: 0 H2O is more nucleophilic

Comparison between hydrogen production via H2S and H2O splitting on transition metal-doped TiO2 (101) surfaces as potential photoelectrodes - ScienceDirect

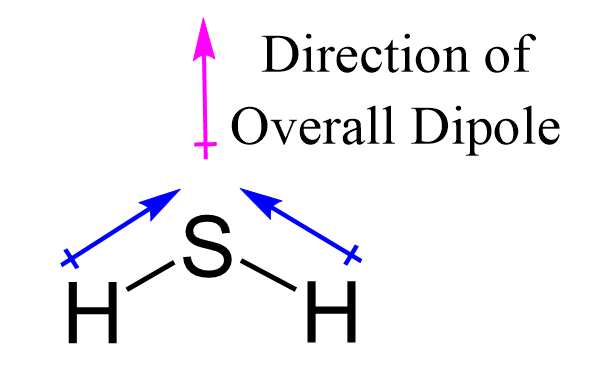
H2O is liquid while H2S is a gas why - Chemistry - The p-Block Elements - 14956293 | Meritnation.com

Hydrate Stability in the H2S–H2O system—Visual Observations and Measurements in a High-Pressure Optical Cell and Thermodynamic Models | Journal of Chemical & Engineering Data

Hydrogen Sulfide, Corrosion and BOD5 in Innovyze InfoSewer – ICM SWMM & ICM InfoWorks, SWMM5 & SWMM5+