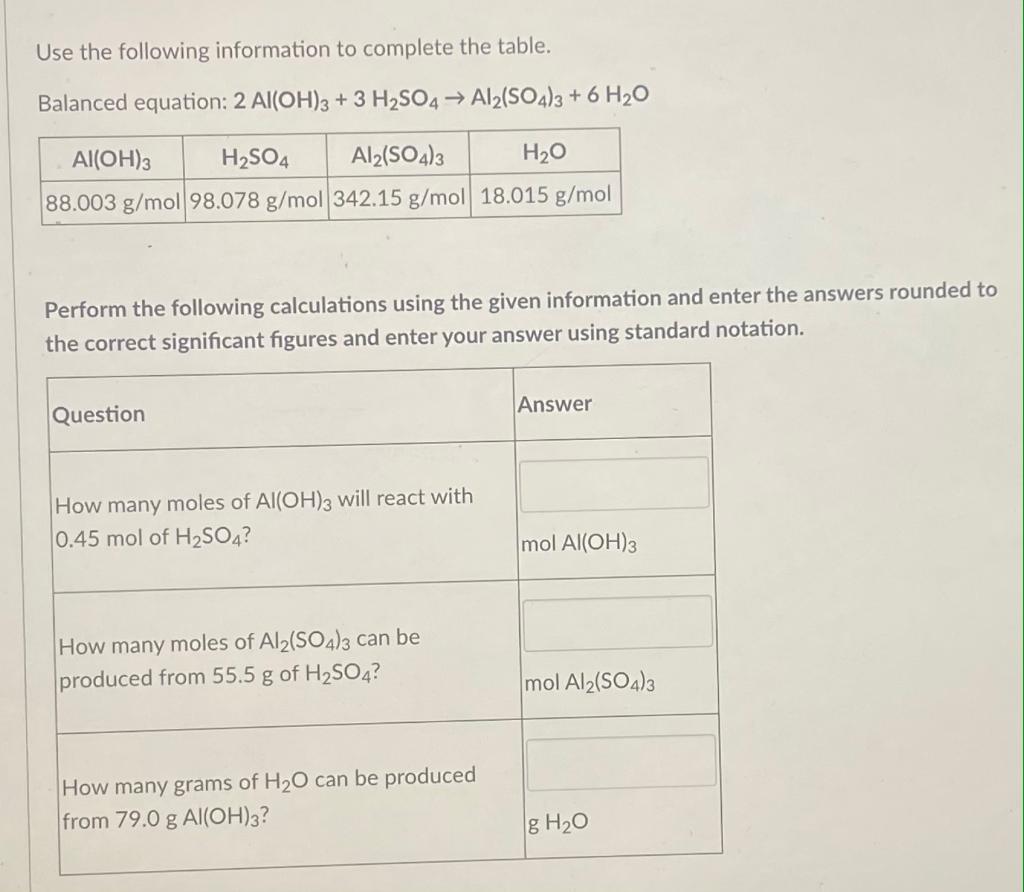
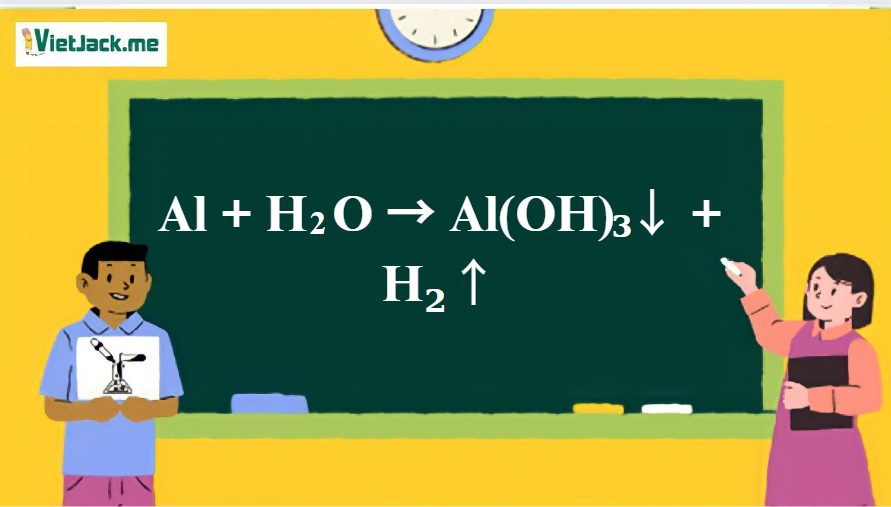Al(H2O)6 3+ + H2O clusters with (A) single H-bond; (B and C) double... | Download Scientific Diagram

SOLVED: Consider the following unbalanced equation: Al4C3 + H2O -> Al(OH)3 + CH4 How many moles of Al(OH)3 will be produced when 0.487 mol of CH4 is formed? 0.649 mol 0.162 mol 0.365 mol 0.487 mol

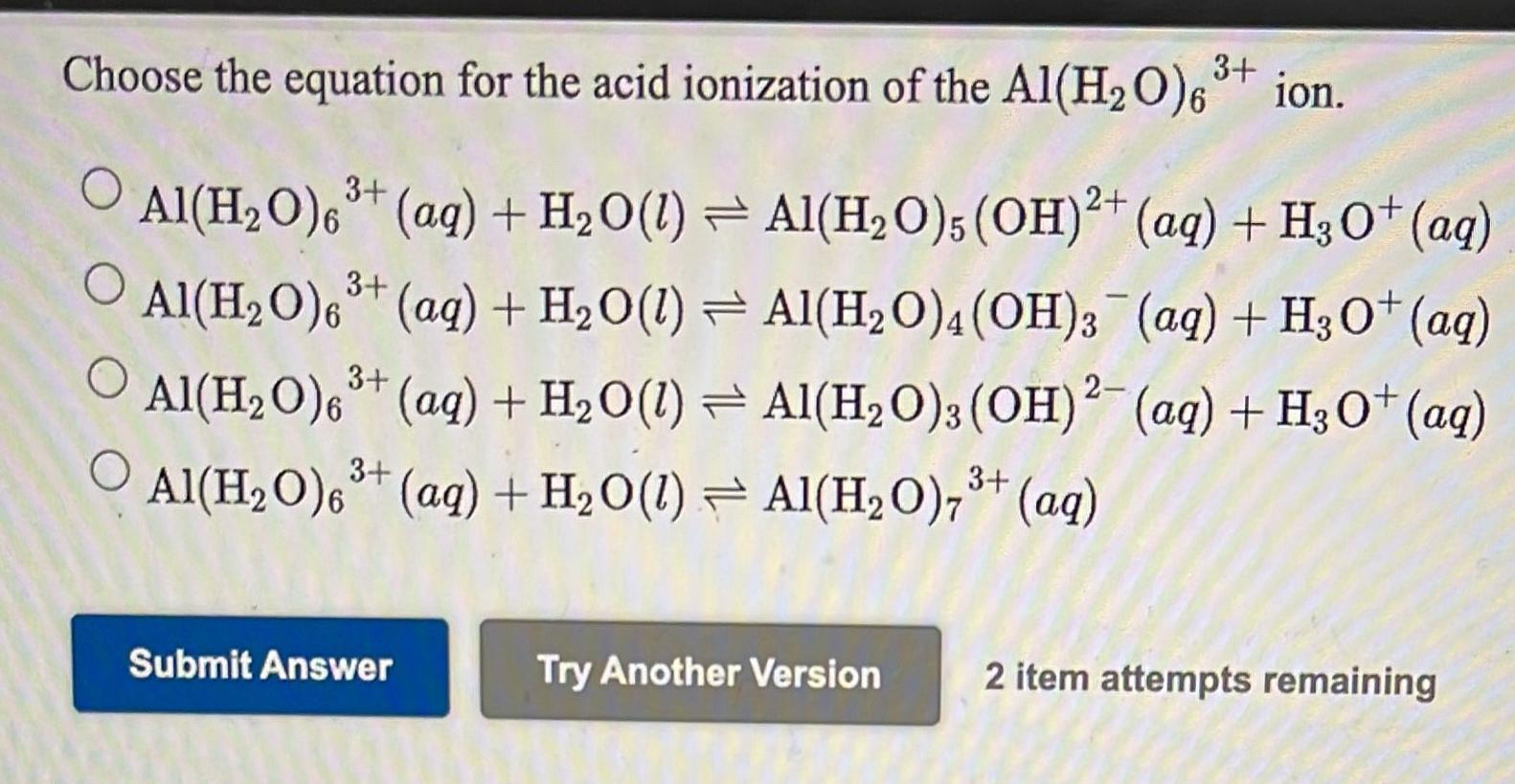
SOLVED: The hydrated Al3+ ion, Al(H2O)6^3+, is a weak acid in water. What are the products of its reaction with H2O? Al(H2O)6^3+(aq) + H2O(l) â†' Al( H2O)5(OH)2^-(aq) + H3O+(aq)
![SOLVED: In the reaction Al3+ + 6H2O → [Al(H2O)6]3+, what does H2O act as? Bronsted-Lowry acid Lewis acid Bronsted-Lowry Base Lewis base SOLVED: In the reaction Al3+ + 6H2O → [Al(H2O)6]3+, what does H2O act as? Bronsted-Lowry acid Lewis acid Bronsted-Lowry Base Lewis base](https://cdn.numerade.com/ask_previews/29288c5c-bf0a-455e-8d4b-2c5553f62277_large.jpg)