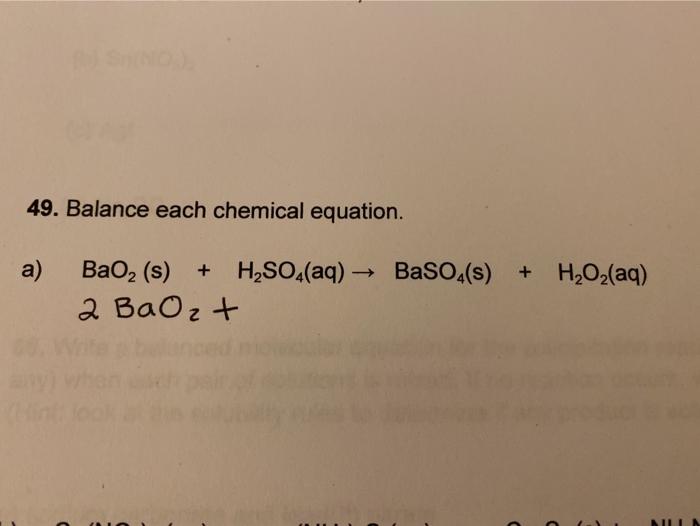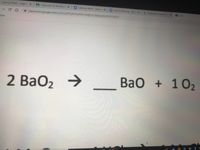Influence of the total pressure on the thermodynamic equilibrium of... | Download Scientific Diagram
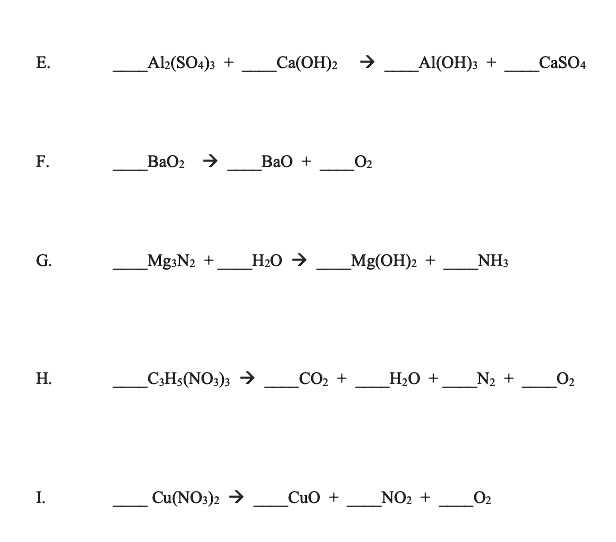
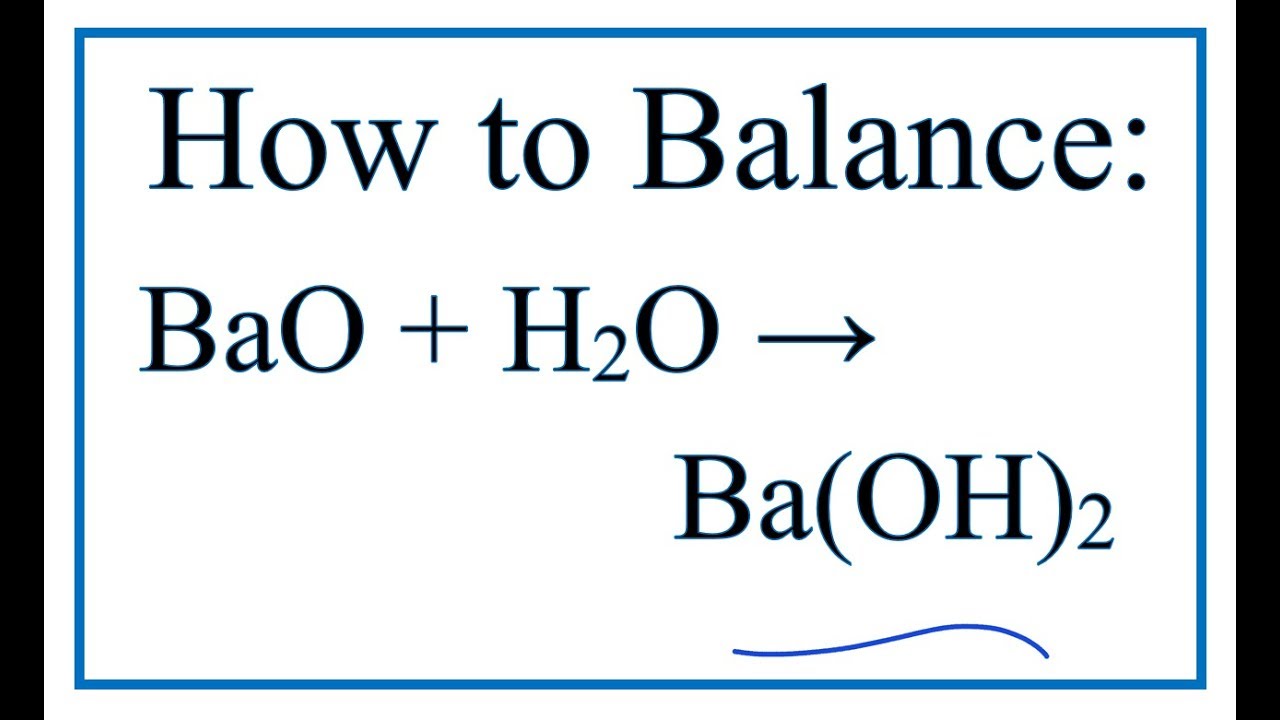
SOLVED: Al2(SO4)3 Ca(OH)2 Al(OH)3 CaSO4 BaO2 BaO Mg3N2 H2O Mg(OH)2 NH3 C2H5(NO3)2 CO2 H2O N2 O2 Cu(NO3)2 CuO NO2

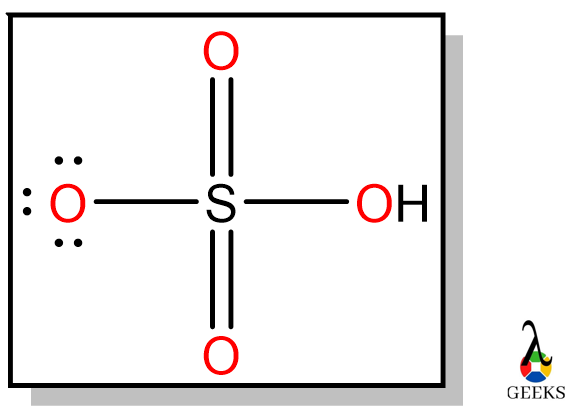
SOLVED: Hydrogen peroxide can be prepared by the reaction of barium peroxide with sulfuric acid according to the reaction: BaO2(s) + H2SO4(aq) â†' BaSO4(s) + H2O(aq) How many milliliters of 2.00 M

The compound that gives \( \mathrm{H}_{2} \mathrm{O}_{2} \) on treatment with dilute \( \mathrm{... - YouTube