1 Bán Hóa chất Sodium dihydrogen phosphate monohydrat, reagent grade - NaH2PO4.H2O - SO0331 - Scharlau giá rẻ ở hcm

Mono Sodium Phosphate Water Treatment Chemicals Nah2po4. H2O - China Monosodium Phosphate, Sodium Dihydrogen Phosphate | Made-in-China.com

ReasonH_3PO_4 is a tribasic acid.AssertionNaOH + H_3PO_4 rightarrow NaH_2PO_4 + H_2O In the given reaction, the equivalent weight of H_{3}PO_{4} is M/3.

SOLVED: A buffer was made by mixing aqueous solutions of NaH2PO4 and Na2HPO4 together. This buffer is made by mixing two salts together. a. Write the balanced dissociation reaction for solid NaH2PO4

Jual sodium dihydrogen phosphate monohydrate merck 1kg / NaH2PO4. H2O - Kota Depok - Leres Mart | Tokopedia
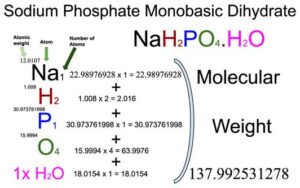
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes

88) The equivalent weight of NaH,PO, in the reaction NaH2PO4 +KOH-NaKHPO4+ H2O is (Given Atomic masses: Na = 23, K = 39, P = 31) (1) 158 (2) 60 (3) 97 (4) 120
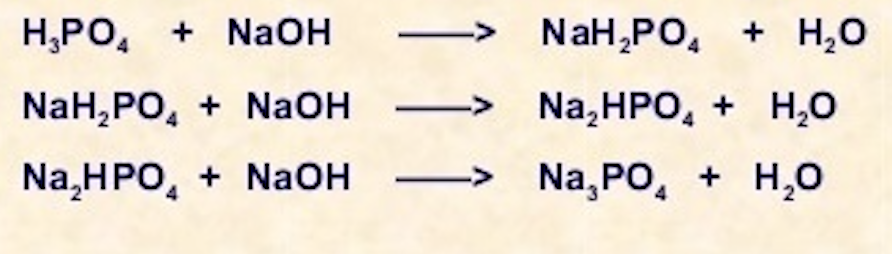
8. Calculate equivalent weight of H3PO4 and Ca(OH)2 on the basis of given reaction H3PO4 + NaOH = NaH2PO4 + H2O Ca(OH)2 + HCl = Ca(OH)Cl + H2O
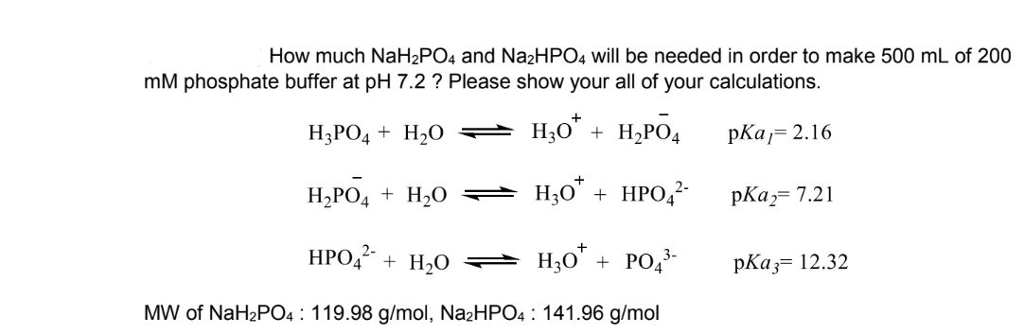
SOLVED: Problem for the group: Determine the weight in grams of sodium dihydrogen phosphate (NaH2PO4.H2O; FW = 138.01) and sodium hydrogen phosphate (Na2HPO4; FW = 141.98) needed to prepare 1L of a
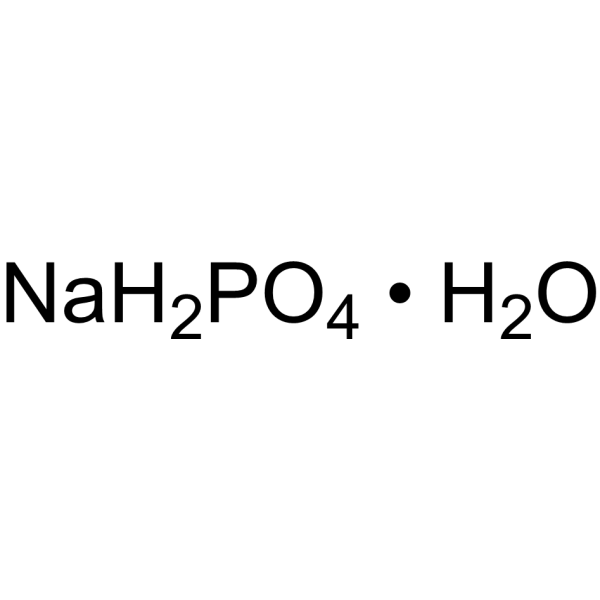
Sodium dihydrogen phosphate monohydrate (Sodium phosphate monobasic monohydrate) | Biochemical Assay Reagent | MedChemExpress

21. The equivalent weight of H3PO, in the given reaction is NaOH + H2PO4 → NaH2PO4 + H2O to Is (1) e 49 enois91 199 (2) 98 (3) 32.67 (4) 24.5 la vso) y diw

Jual NaH2PO4 H2O Sodium Dihydrogen Phosphate Monohydrate Ecer 10gram - Kota Makassar - Sentanasempurna | Tokopedia
Sodium Dihydrogen Phosphate Starting From Sodium Chloride and Orthophosphoric Acid Via Cation Resin Exchange

Welcome to Chem Zipper.com......: In a determination of P an aqueous solution of NaH2PO4 is treated with a mixture of Ammonium and magnesium ions to precipitate magnesium Ammonium phosphate (Mg(NH4)PO4.6H2O. This is