Calculate the % of boron (B) in borax Na2 B4 O7 . 10H2O. (H = 1, B = 11, O = 16, Na = 23) - Sarthaks eConnect | Largest Online Education Community

BORON & ITS COMPOUNDS 2. By passing electric discharge at low pressure through a mixture of BCl 3 or BBr 3 and excess of hydrogen. 3. By reacting. - ppt download

Hydrated sodium tetraborate is heated to drive off the water. You find there are 0.01312 mol Na2B4O7 and - brainly.com
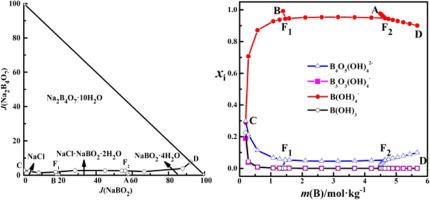
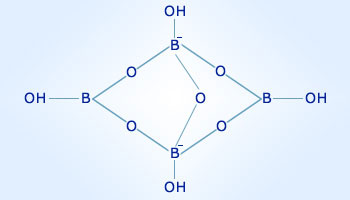
Solubility determination and thermodynamic modelling of solid−liquid equilibria in the (NaCl + NaBO2 + Na2B4O7 + H2O) system at 298.15 K,The Journal of Chemical Thermodynamics - X-MOL


.png)




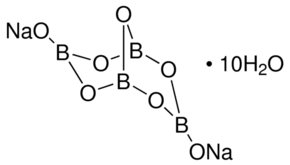



![Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA](https://t.pimg.jp/076/297/336/1/76297336.jpg)




